 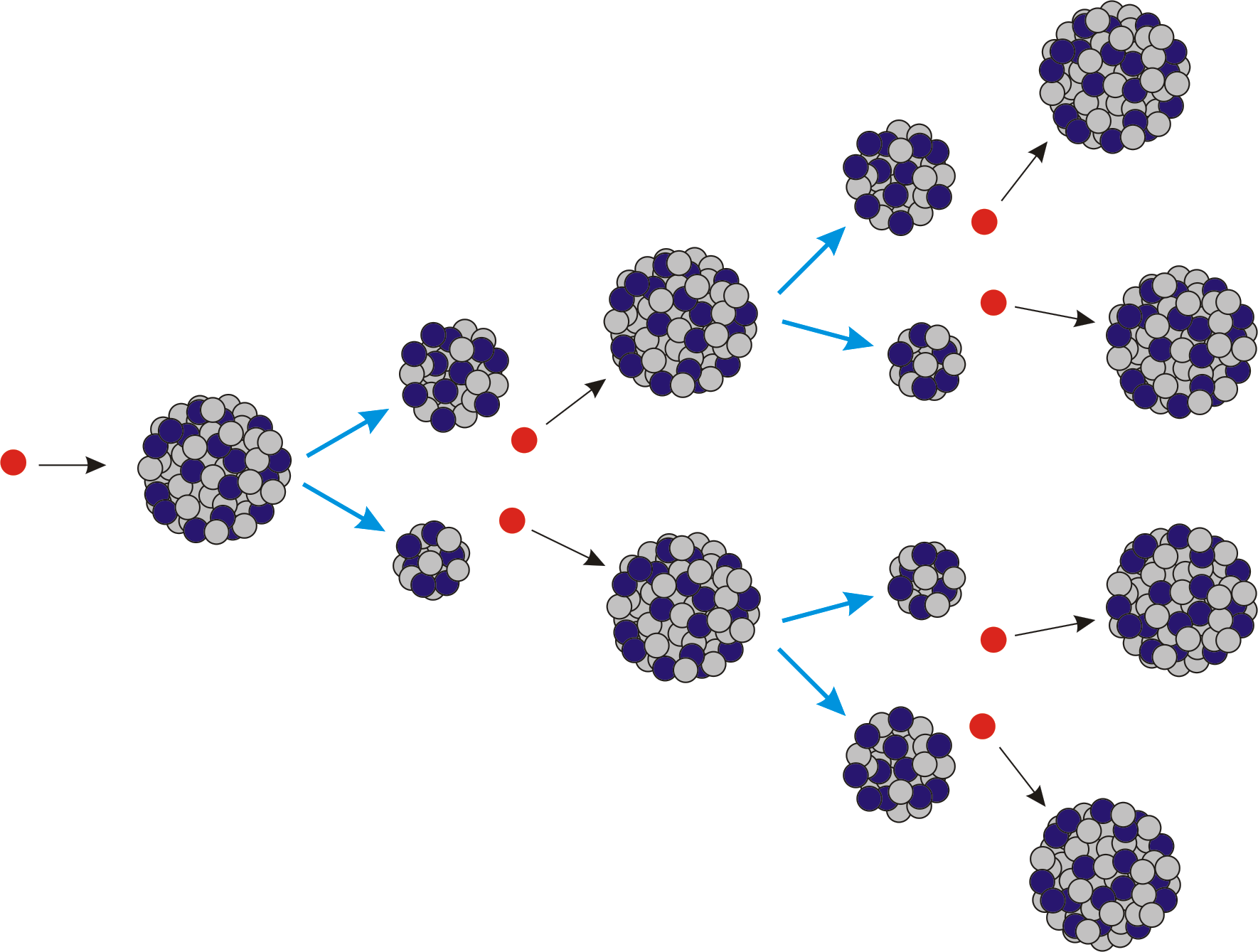
The large amounts of heat released by fission reactions must be tightly controlled. The reactor core is surrounded by thick walls of concrete and steel to help shield the reaction from the outside world and the world from the reaction, especially in the event of a meltdown. Control rods made of neutron-absorbing materials (e.g., cadmium, hafnium, boron) can be inserted or withdrawn from the reactor core to adjust the rate of the reaction. Fuel rods are submerged in a circulating water bath to prevent overheating. The energy released in the fission reaction is used to boil water into steam that spins turbines to produce electricity. These fuel rods contain small pellets of fuel clad in a corrosion-resistant zirconium alloy that allows neutrons to pass through. In nuclear power plants, nuclear fuel is typically shaped as rods, which are arranged in the nuclear reactor core. How to control a chain reactionĬontrolled chain reactions, which are used in nuclear power plants and many medical applications, employ several methods to slow down or absorb the neutrons produced by nuclear fission, which in turn slows the fission rate until the reaction eventually stops. Chain reactions have the possibility of releasing enormous amounts of energy, but can also run out of control. If a “critical mass” of nuclear fuel is used, the chain reaction can become self-sustaining. If these neutrons collide with other unstable nuclei, they can create a chain reaction that continues the nuclear fission process. The splitting of a large nucleus releases multiple lighter nuclei, a large amount of energy, and free neutrons. The nuclei of some large atoms like uranium, thorium, and plutonium are unstable and can be split when bombarded with neutrons. 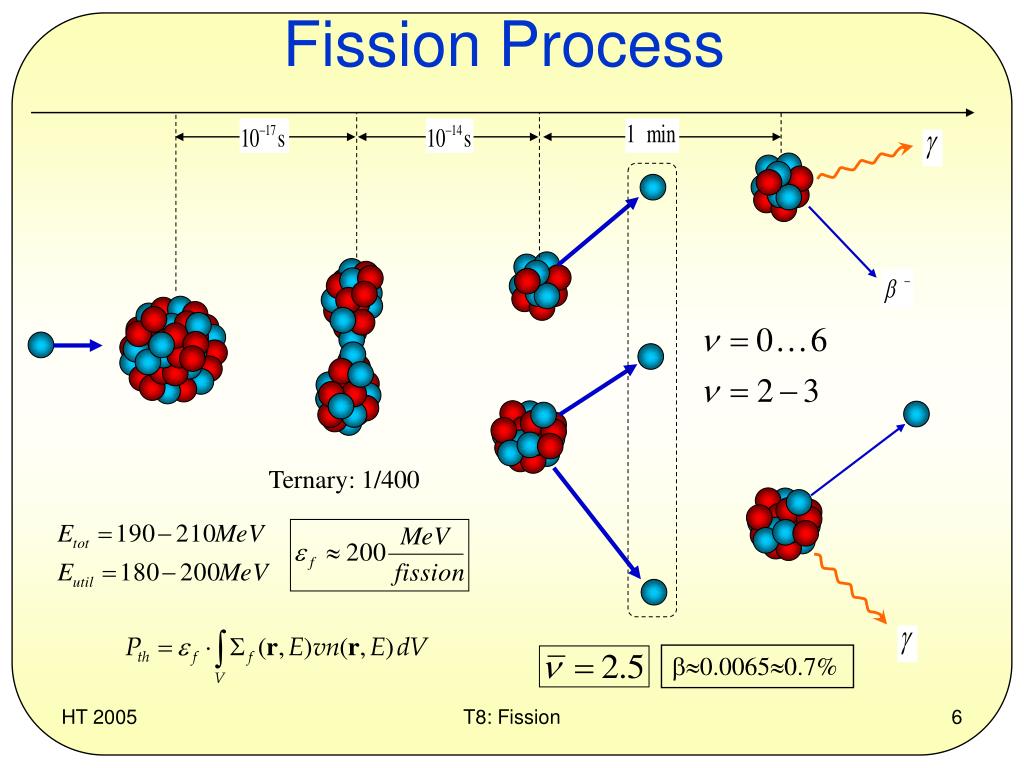
Nuclear fission, which powers nuclear reactors, is the splitting of nuclei. In a fusion reaction, the nuclei of two atoms are joined together to create a heavier atom. There are two main types of nuclear reactions: fusion and fission. Many elements have multiple isotopes, and these vary in their stability.įigure 1. The number of neutrons defines the isotope number. 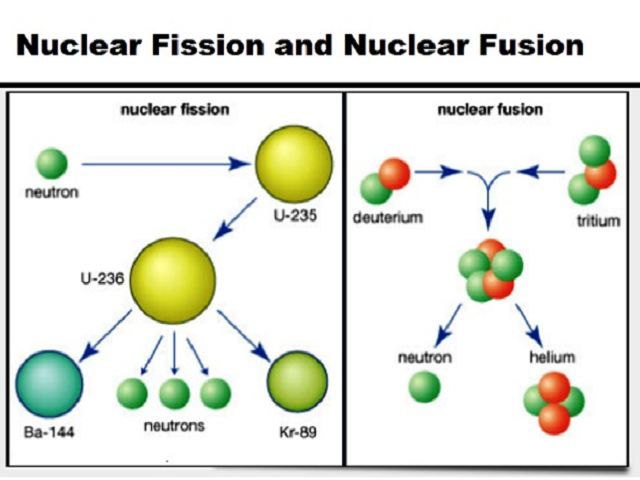
The number of protons defines the atomic number, which is distinct for each element. The nucleus (plural, nuclei) is surrounded by a cloud of electrons, which contribute very little to the overall mass of an atom. Atoms contain a dense nucleus composed of protons and, in all cases but hydrogen, neutrons (Figure 1). Basics of nuclear energyĪll matter is composed of atoms. However, the 25th anniversary of the Chernobyl disaster and the recent and evolving Fukushima Daiichi disaster serve as reminders that the benefits of nuclear power do not come without risks. This electricity comes from a seemingly ideal source: nuclear power is cost-effective, does not rely on fossil fuels, and emits the same carbon equivalent per kilowatt-hour as wind and hydropower. Today, twenty-nine countries operate nuclear power plants, and these produce about 14% of the world’s electricity. On December 20 th, 1951, four glowing light bulbs in Arco, Idaho heralded the first use of nuclear power for electricity generation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
